TBR achieves EU-MDR certification for all its implants
PRESS RELEASE – Toulouse, April 7th, 2026
TBR Dental Group announces the CE marking under the European Medical Device Regulation (EU MDR) 2017/745 for all its implants.
This certification, one of the most demanding in the world, confirms the quality, safety and performance of TBR’s implant solutions, as well as the robustness of its industrial and regulatory processes.
“This certification marks a key milestone for TBR. It validates our scientific rigor and strengthens our ambition to set a new standard of prevention in implantology,” says Julien Benhamou, President.
“We congratulate all our teams on the work accomplished and for this collective achievement that reflects our commitment to safety and reliability, both for dentists and patients ,” says Dr. Olivier Benhamou, Chief Scientific Officer.
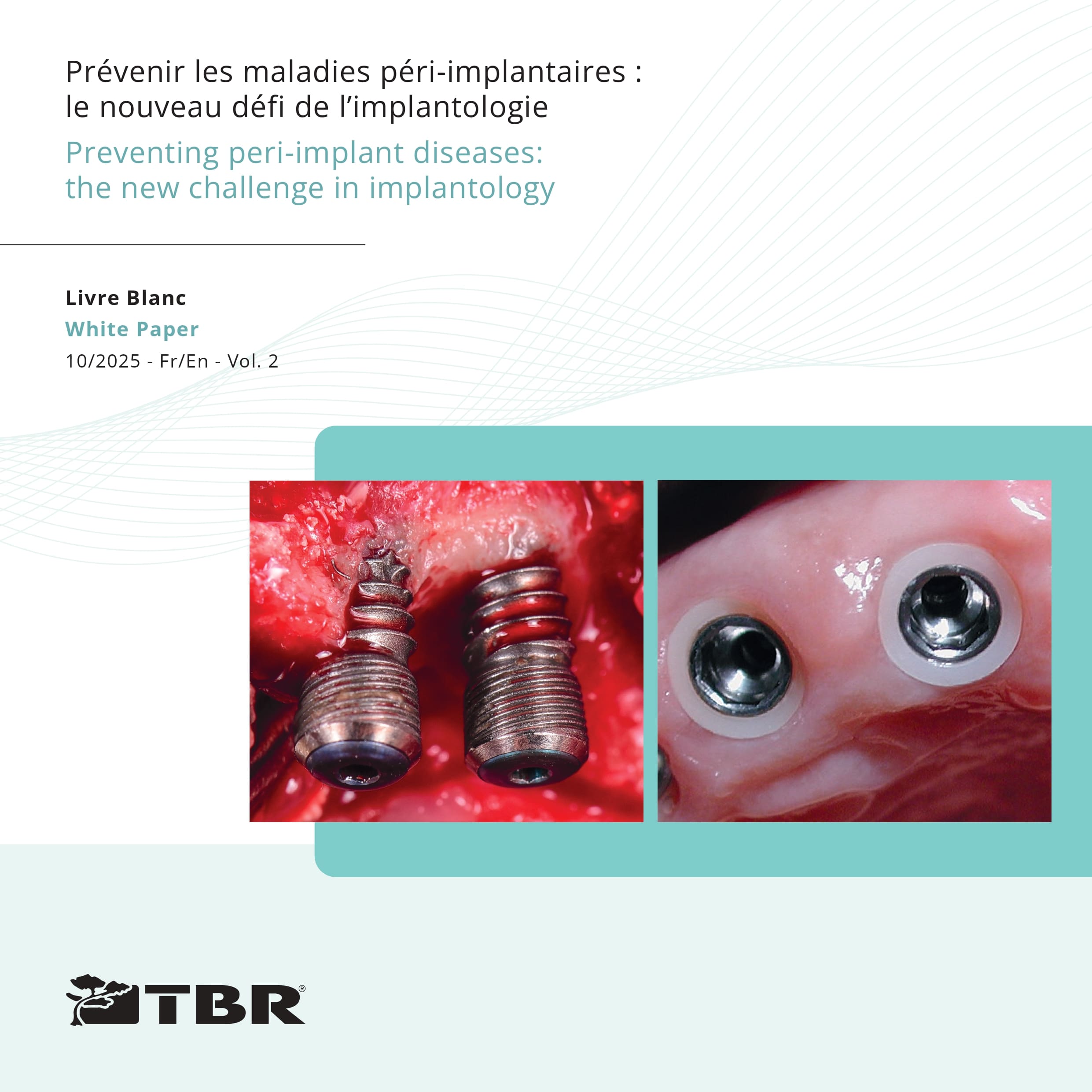
The Z1 implant, with its patented zirconia collar, fully illustrates this commitment with a preventive approach to implantology, aiming to preserve the biological width and limit peri-implant complications as detailed in the White Paper “Preventing peri-implant diseases: the new challenge in implantology”.
With this recognition, TBR reaches a new level of excellence and approaches the future with confidence, continuing its efforts in innovation and international development.
About TBR Dental Group:
A French leader in implantology since 1987, TBR is present in over 40 countries and develops innovative and preventive solutions serving clinicians and patients.